 Keeping in mind that the association between a metal and a non-metal usually results in developing an ionic compound. As gaseous ions combine to form atoms, the energy required for the conversion value is expressed as a kJ/mol unit. Since the reaction is exothermic in certain conditions, the value of the lattice energy is always negative. While we expect a systematic system to be less unchanging since its entropy is limited, the situation is not the same. Second, this energy is used in the reverse phase in which gaseous ions bond to form the atoms of an ionic solid. This description gives meaning to the lattice energy, which is always positive because the reaction is endothermic. One is the energy necessary for breaking an ionic solid as its atoms are converted to ions (gaseous). Lattice Energy definition can be explained in two distinct ways by a sort of potential energy. Generally, it is expressed as kilojoules per mole (kJ/mol). Even so, it can be calculated with the help of the Born-Haber cycle. In an Ionic solid, lattice energy cannot be directly measured. It gives insights into various characteristics, including its volatility, solubility, and durability of ionic solids. Also, it can be described as a method of measuring cohesive forces that bind ions. Lattice energy is a calculation of ionic bond strength in an ionic compound.  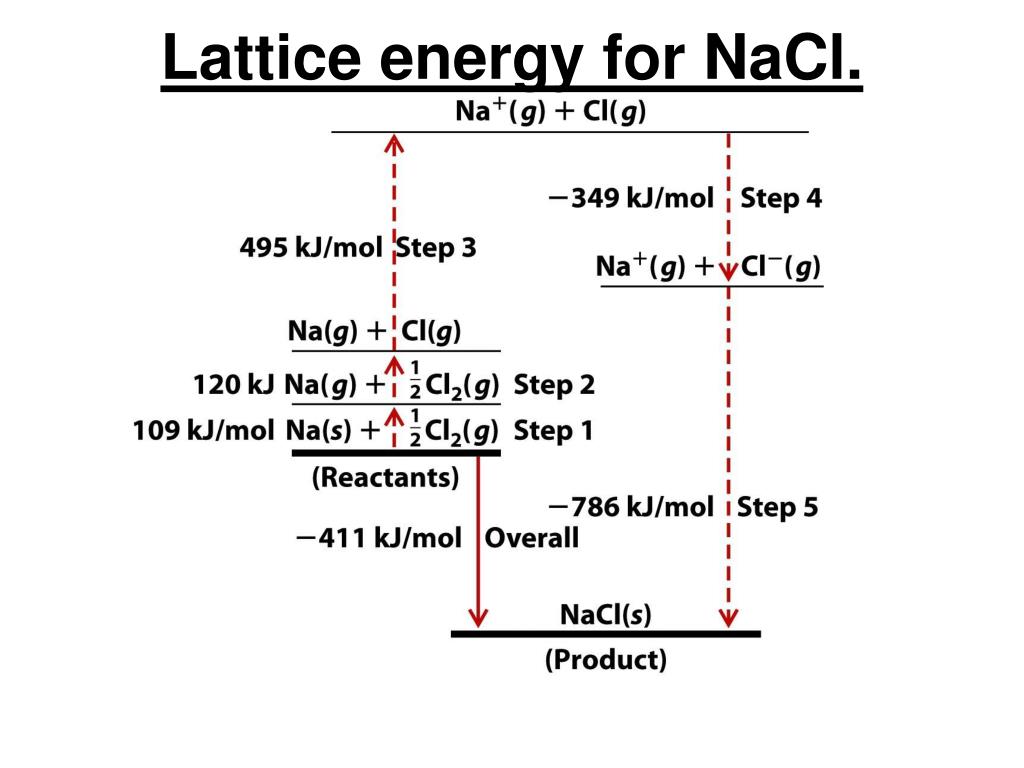
This article will elaborate only on lattice Energy, Lattice Energy definition, Lattice energy formulas, Lattice Energy equation, and some practical examples to understand Lattice Energy’s concepts. On the other hand, Lattice Enthalpy is considered the change in enthalpy whenever the bond is formed or broken. Lattice energy is the amount of energy that is required to break a solid bond into gaseous elements. However, the difference between them is negligible. These terms are differentiated based on their mode of calculation. The fact is that Lattice energy and Lattice Enthalpy has a different meaning. Both are used to calculate bond strength in an ionic or Covalent bond that ultimately elaborates the attractive forces in ions, so these terms are problematic. Another way round, some Students Consider it, they have a different meaning. Some students might consider it they have the same meaning. Cartoon by Nick D Kim (, used by permission).In Chemistry, Lattice energy and Lattice enthalpy are the two most commonly used terminologies. 
The chemistry of potassium is dominated by electron loss to form K +. 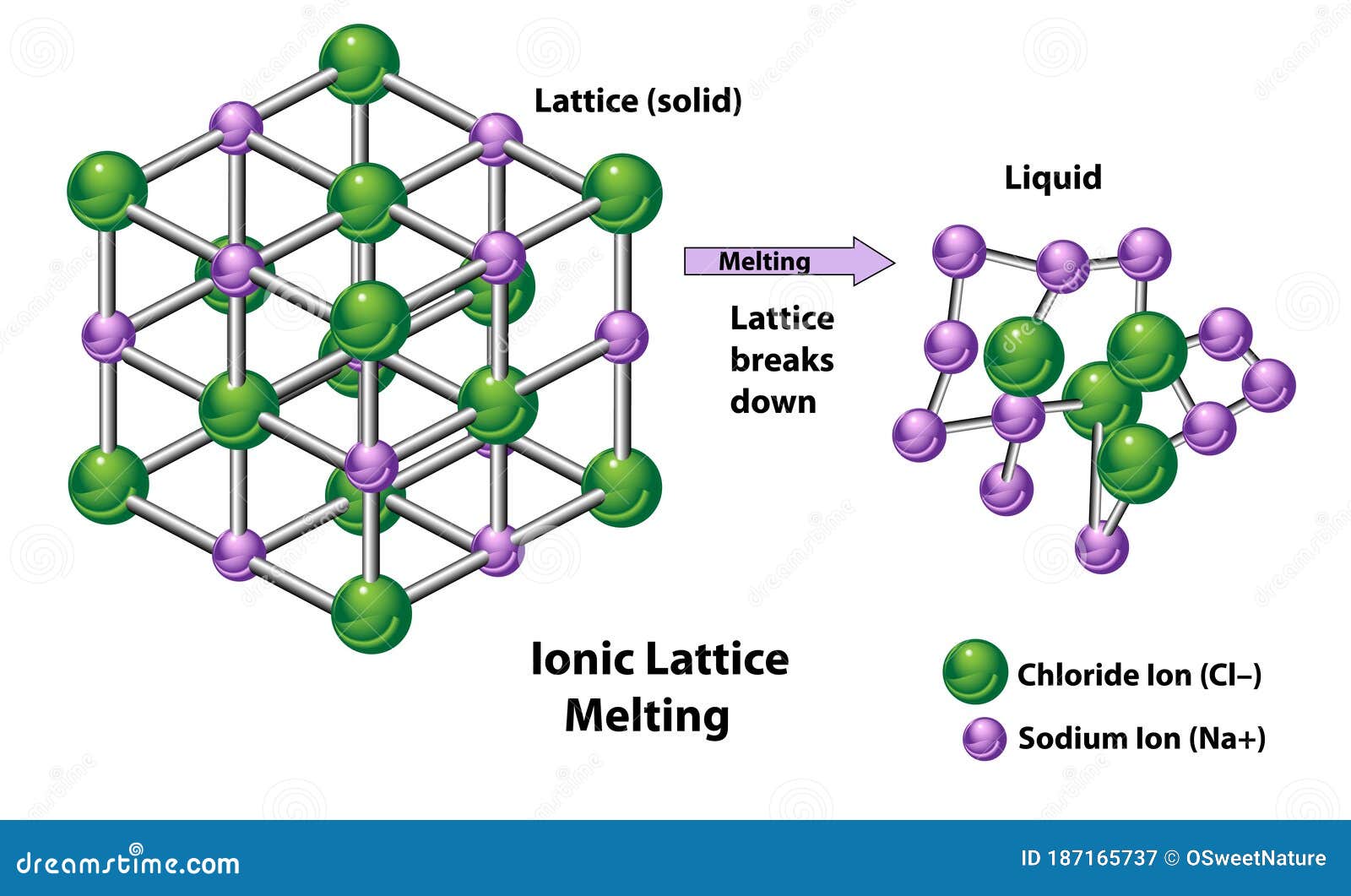
The picture above shows the colour arising from a burning mixture of potassium chlorate (KClO 3) and sucrose (only to be demonstrated by a professionally qualified chemist). Potassium and its salts impart a lilac colour to flames. It usually catches fire during the reaction with water. As do all the other metals of the alkali group, it decomposes in water with the evolution of hydrogen. It oxidises very rapidly in air and must be stored under argon or under a suitable mineral oil. It is silvery in appearance immediately after a fresh surface is exposed. It is one of the most reactive and electropositive of metals and, apart from lithium, it is the least dense known metal. Potassium is never found free in nature, but is obtained by electrolysis of the chloride or hydroxide, much in the same manner as prepared by Davy. It is also a vital element in the human diet. Potassium is an essential constituent for plant growth and it is found in most soils. Potassium is a metal and is the seventh most abundant and makes up about 1.5 % by weight of the earth's crust. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
